March 21, 2026
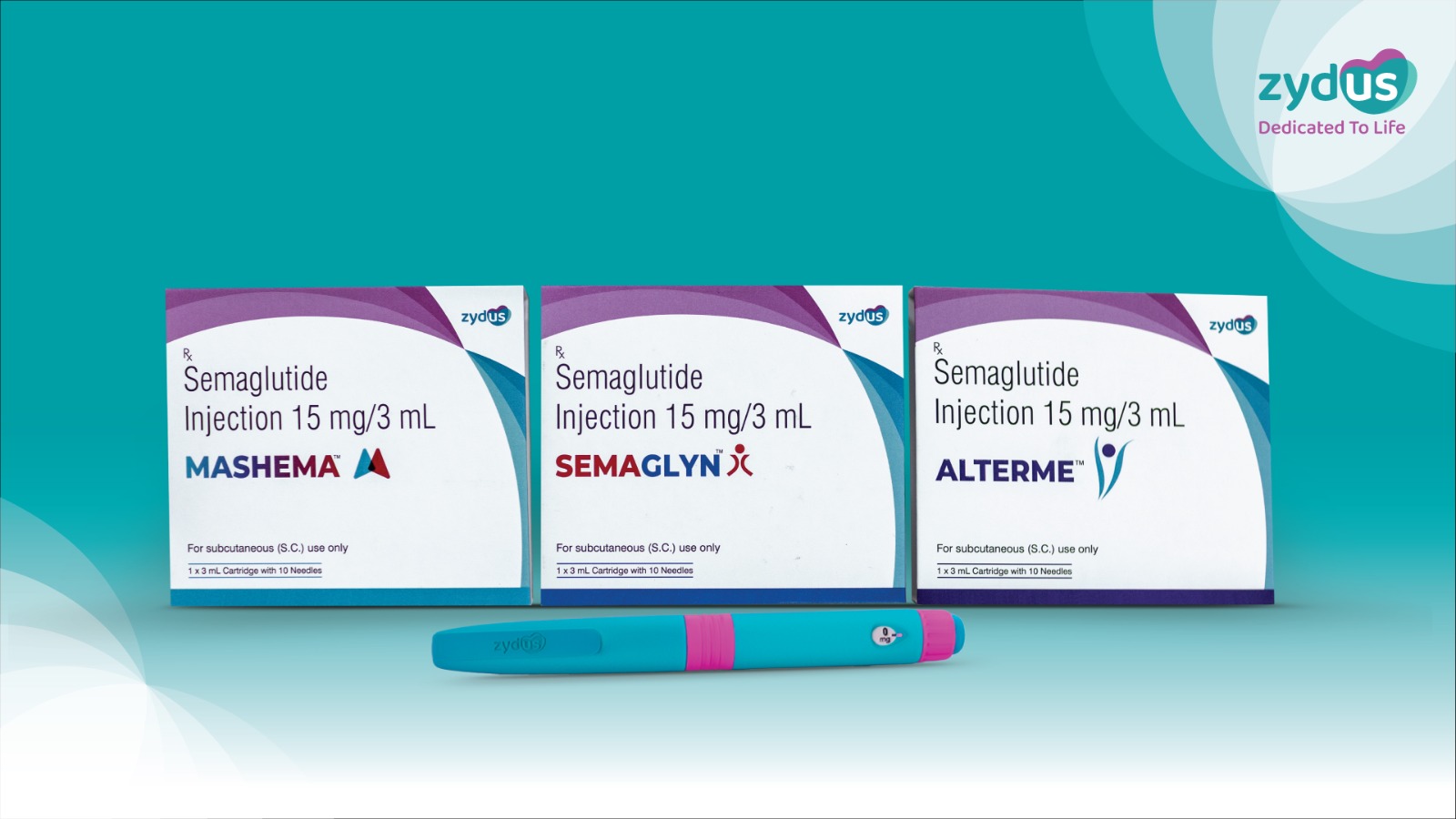
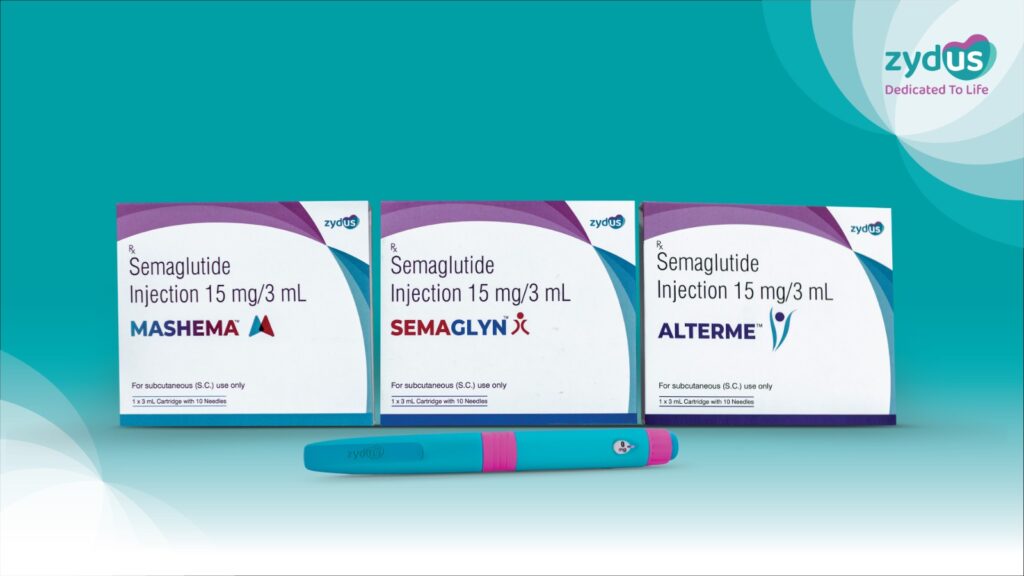
Zydus Lifesciences Limited has announced the launch of its Semaglutide Injection in India under the brand names SEMAGLYN™, MASHEMA™ and ALTERME™, following patent expiry in the country. The approval for manufacturing and marketing the drug for Type 2 Diabetes Mellitus and Obesity was granted earlier by the Drug Controller General of India (DCGI).
In a significant innovation, Zydus has introduced the therapy through a reusable multi-dose pen device, a first-of-its-kind offering in this category in India. Unlike conventional single-use pens, the new device uses a prefilled cartridge (15 mg/3 ml), allowing patients to adjust and administer varying doses from a single pen. This approach is expected to improve treatment adherence, enhance convenience, and substantially reduce therapy costs.
The company holds exclusive rights for this patient-friendly delivery system, which is designed to simplify long-term treatment. The injection will be manufactured at Zydus Biotech Park in Ahmedabad, reinforcing the company’s domestic production capabilities.
Zydus has indicated that the average monthly cost of therapy will be around ₹2,200, making it a more accessible option for patients compared to existing alternatives.
India continues to face a growing burden of metabolic disorders. According to global estimates, nearly 89 million adults in the country are living with diabetes, accounting for about 10.5% of the adult population. At the same time, obesity rates are rising sharply, with significant increases observed across both men and women in recent years.
Experts believe that GLP-1 based therapies, such as Semaglutide, can play a transformative role in managing both diabetes and obesity, addressing one of India’s most pressing public health challenges.